
Rikako Kato
Researcher (Amemori-G)
- Position
- Assistant Professor
- Research Field
- Neurophysiology
Research Overview
Decision making and emotions
In the modern world, “His decision was strongly influenced by emotion” has a negative connotation. People think that emotions are at odds with calm, rational decisions. But, the fact that rich emotions have survived long evolutionary timescales implies that emotions are in some way useful for survival or conservation of a species. My research aims to specifically reconcile the negative image of emotions with their true functional roles in survival. Based on this, I study decision making based on action values calculated by reward prediction errors encoded in dopamine neurons. Action values represent the brain’s weighting of decisions based on how similar decisions triggered rewards in the past (Fig.1). The goal is to achieve the largest payout (reward) and to minimize any punishment. Predicting the rewards that will result from an action is a major factor in decision making. Interestingly, the optimal action is not always selected for a given situation. However, this is not a mistake. Rather, it is due to the brain calculating future profits based on a wider context than simple reward prediction. For example, sometimes it is advantageous to explore to acquire novel information, or to change your strategy by more aware of your environment. One function of emotion is to influence the weighting of action value. For example, anger triggers an approach to negative events, such as a struggle with an enemy. Previous studies suggested that brain areas such as the anterior cingulate cortex (ACC), the nucleus accumbens (NAc) and the bed nucleus of the stria terminalis (BNST) are hubs for bridging between reward value information and emotional valence information. However, we are not yet sure how neural activity in these parts of the brain is correlated representing emotion itself and balancing between different type of emotions, especially in humans and other primates.
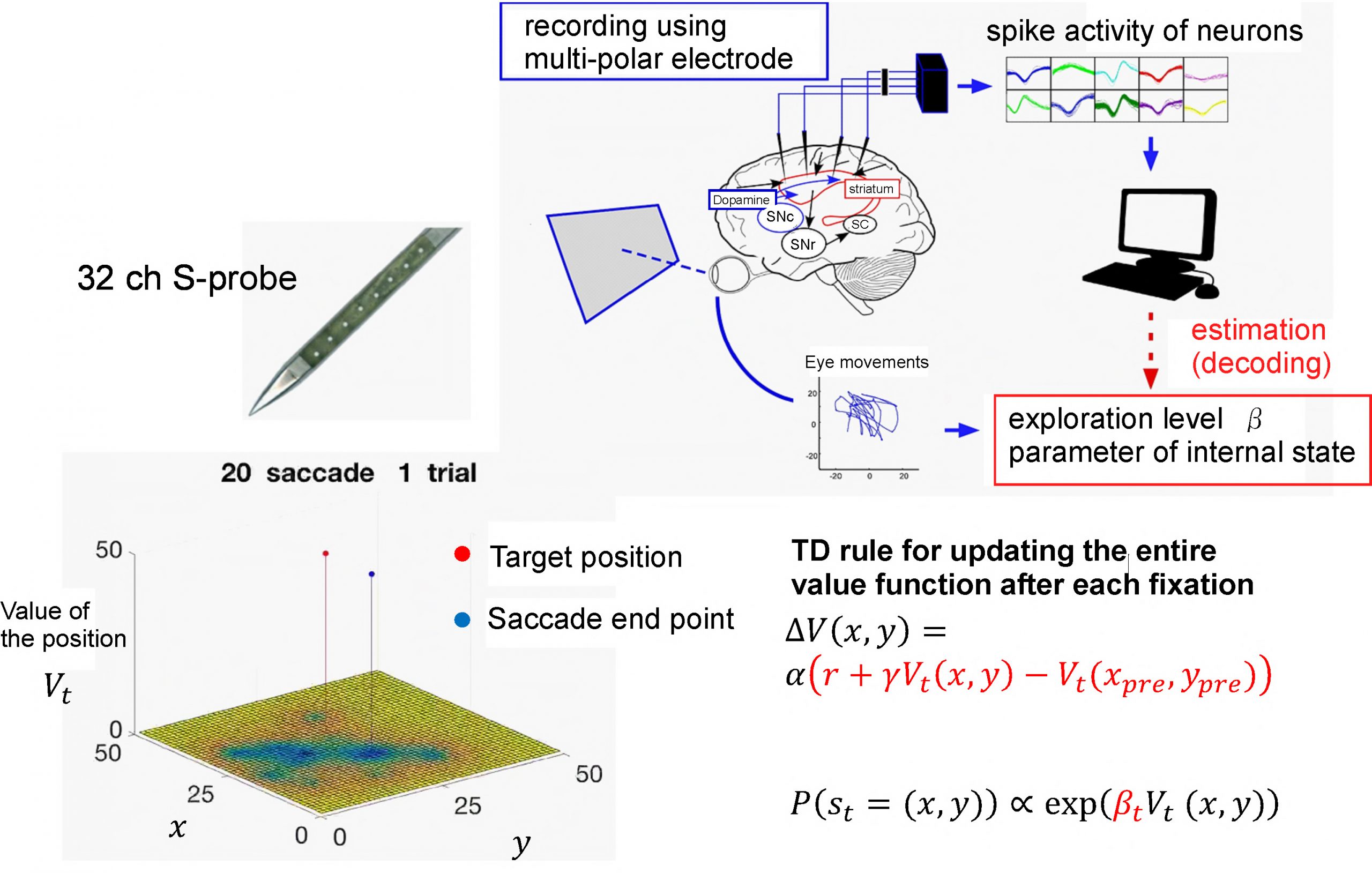
Fig.1 Reinforcement learning model and Neural decoding of decision making.
Biography
Rikako Kato obtained her PhD (Physiology) from Tsukuba University (2003). She was appointed as an postdoctral fellow of Collège de France (Laboratoire de Physiologie de la Perception et de l’Action) and a temporary teaching and research associate (2003-2005). Then she was appointed as a program-specific researcher (CREST or Brain Sciences) at National Institute for Physiological Sciences (NIPS, 2005-2012), a researcher at NIPS (2012-2016) and a program-specific researcher (AMED) at Department of Neuroscience, Graduate School of Medicine, Kyoto University (2016-2020). In 2021, she moved to ASHBi.
Publications
加藤利佳子 盲視:第一次視覚野損傷下での視覚意識を伴わない急速眼球運動調節機構, Kato R.,Blindsight : Saccadic Eye Movement Control Mechanisms Without Visual Awareness in Primary Visual Cortex Lesions 「神経眼科」第41巻 第1号 別冊 Neuro-ophthalmology Japan Vol.41. No.1.
Kato R, Zeghbib, A., Redgrave, P., and Isa, T. (2021) Visual instrumental learning in blindsight monkeys. Sci Rep. 11(1):14819
Kato, R., Hayashi, T., Onoe, K., Yoshida, M., Tsukada, H., Onoe, H., Isa, T., and Ikeda, T. (2021) The posterior parietal cortex contributes to visuomotor processing for saccades in blindsight macaques. Commn. Biology. (4), 278
Kinoshita, M., Kato, R., Isa, K., Isa., Kobayashi, Kenta, Kobayashi, Kazuto, Onoe, H., and Isa, T. (2019) Dissecting the circuit for blindsight to reveal the critical role of pulvinar and superior colliculus. Nat Commun. 10(1):135.
Takakuwa, N., Kato, R., Redgrave, P and Isa, T. (2017) Emergence of visually-evoked reward expectation signals in dopamine neurons via the superior colliculus in V1 lesioned monkeys. Elife 6. Pii: e24459.
Yoshida, M., Itti, L., Berg, DJ., Ikeda, T., Kato, R., Takaura, K., White, BJ., Munoz, DP., and Isa, T. (2012) Residual attention guidance in blindsight monkeys watching complex natura scenes. Curr. Biol. 22(15): 1429-34.
Kato, R., Takaura, K., Ikeda, T., Yoshida, M., and Isa, T. (2011) Contribution of the retino-tectal pathway to visually guided saccades after lesion of the primary visual cortex in monkeys. Eur. J. Neurosci. 33(11):1952-60.
Yoshida, M., Takakuwa. K., Kato, R., Ikeda, T., and Isa, T. (2008) Striate cortical lesions affect deliberate decision and control of saccade: implication for blindsight. J. Neurosci. 28(42):10517-30.
Kato, R., Grantyn, A., Dalezios, Y., and Moschovakis, AK. (2006). The local loop of the saccadic system closes downstream of the superior colliculus. Neuroscience. 143(1):319-37.
Kato, R., Iwamoto, Y., and Yoshida, K. (2003). Contribution of GABAergic inhibition to the responses of secondary vestibular neurons to head rotation in the rat. Neurosci. Res. 46(4):499-508.
Awards
F1000 Prime, Article Recommendations (2012.4)
Research Group
Joined
Apr. 1, 2021
