研究概要
Investigating the changes in eye movement behavior as an early diagnostic biomarker for Parkinson’s disease using marmosets as the animal model.
The oculomotor system reflects the cognitive state and is widely used to probe neural disorders in human. Despite the distinctive phenotypical features associated with each neural disorder, almost all share an alteration in the oculomotor function due to its vulnerability to the toxic protein aggregates, among other reasons.
Parkinson’s disease (PD) is neurodegenerative disease characterized by death of dopaminergic neurons of the substantia nigra, along with accumulation of α-Synuclein (α-Syn) containing inclusion bodies (Lewy pathology). This results in a characteristic motor impairment that is usually seen only when more than 70% of the dopaminergic neurons are already dead and it is too late to treat the patients. In addition, there are also non motor symptoms such as neuropsychiatric symptoms, eye movement irregularities and sleep abnormalities. Because eye movement is relatively a direct indicator of the basal ganglia (BG) output, saccade recordings is one of the most insightful tools for various neurological disorders, in particular BG disorders.
The dual- hit hypothesis proposed by Braak suggested that PD is initiated from the gut or the olfactory bulb (OB) and from there, it propagates via the vagal nerve and the OB to the entire CNS.
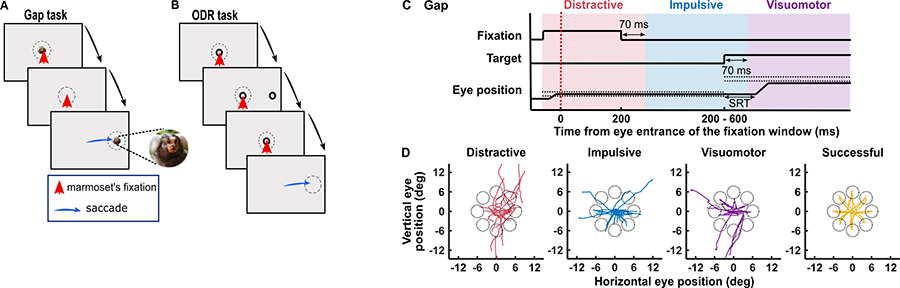
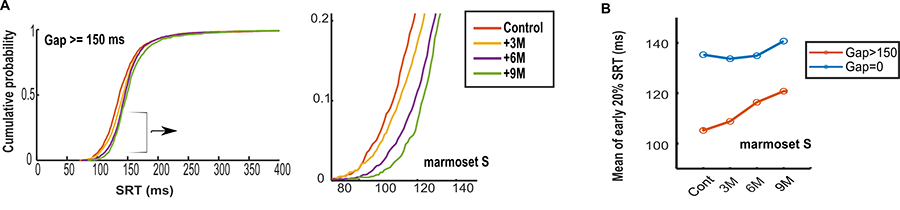
My research focus on studying the longitudinal changes in eye movement behavior and the error answers using marmosets as PD model. To do so, marmosets are trained to perform two different visual tasks, gap, and oculomotor delayed response (ODR) task “<Figure 1>”. By comparing the control and the disease state as the disease progress, any changes in eye movement behavior (saccade reaction time (SRT), endpoint accuracy, curvature, main sequence, …) can be detected. One finding we saw was a gradual decrease in SRT (slower response) with disease progression, “<Figure 2>”.
I’m planning to further expand this and study other visual paradigm and motor movement behavior analysis.
略歴
Wajd Amly joined the Neuroscience Ph.D. course at Kyoto University (2018). She studied the longitudinal changes in eye movement behavior in synucleinopathies, mainly Parkinson’s disease, using common marmosets as an animal model. She is now a researcher in ASHBi.
論文
Amly, W. and Karaman, R. 2016. Overview on the recent drugs delivery approaches. Aperito Journal of Pharmacology and Drug Design, 2. (http://dx.doi.org/10.14437/23788968-2-114)
Amly, W. and Karaman, R. 2016. Recent updates in utilizing prodrugs in drug delivery (2013-2015). Expert Opinion Drug Delivery, 13, 571-91.
Amly, W. and Karaman, R. 2015. Lipid lowering medications, uses, side effects, pharmacokinetic properties, and approaches to improve bioavailability “Commonly Used Drugs - Uses, Side Effects, and Approaches to Improve Its Bioavailability. Nova science publishers.
Amly, W. and Karaman, R. 2014. Antibody directed enzyme prodrug therapy (ADEPT) “Prodrugs Design – A New Era”. Nova science publishers.
Karaman, R., Amly, W., Scrano, L., Mecca, G. and Bufo, S. A. 2013. Computationally designed prodrugs of statins based on Kirby’s enzyme model. J Mol Model, 19, 3969-82.




